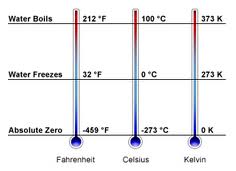
Absolute Zero: On the Kelvin scale, the number 0 is assigned to the lowest possible temperature.

Bimetallic Strip: Two strips of different metals, say one brass and the other of iron, are welded or riveted together.

Calorie: the most common use unit for heat

Celsius Scale: the gap on a scale between freezing and boiling is divided into 100 equal parts, called degrees.

Fahrenheit Scale: On the temperature scale used commonly in the U.S, the number 32 designates the temperature at which water freezes, and the number 212 is assigned to the temperature at which water boils.

Heat: the energy that transfers from one abject to another because of a temperature difference between them.

Internal Energy: the grand total of all energies inside a substance.

Kelvin Scale: the scale used in scientific research is the SI scale.

Kilocalorie: is 1000 calories ( the heat required to raise the temperature of 1 kilogram of water by 1.c)

Specific Heat Capacity: water absorbs more heat per gram then iron for the same change in temperature.

Temperature: the quantity that tells how hot or cold something is compared with a standard.

Thermal Contact: when heat flows from one object or substances

Thermal Equilibrium: after objects in thermal contact with each other reach the same temperature, no heat flows between them

Thermostat: a practical application of a bimetallic
